Septic Arthritis of the Native Shoulder in a Healthy Adult
Noah Quinlan, BA, Shivam Upadhyaya, BA, Brian A. Mosier, MD, Scott D. Martin, MD
The authors report no conflict of interest related to this work.
©2016 by The Orthopaedic Journal at Harvard Medical School
Septic arthritis of the native shoulder joint is a rare condition that can lead to serious sequelae, including death, if not treated acutely. Septic arthritis should be considered in patients presenting with joint pain and decreased range of motion when an alternate diagnosis is not readily apparent. In the absence of known risk factors, septic arthritis is exceedingly rare. Although uncommon, spontaneous septic arthritis can occur in an otherwise healthy patient as there are reports documented in the literature. Here we report one such case in the glenohumeral joint of a healthy 62-year-old woman. We explore the etiology, diagnostic steps, management, and sequelae of spontaneous septic glenohumeral arthritis in an otherwise healthy patient, as well as review the literature.
Septic arthritis of the native glenohumeral joint is a rare condition with serious sequelae if not diagnosed and treated appropriately. Overall, the incidence of septic arthritis has been reported as 4-10 per 100,000 patient-years per year with approximately 11% of cases resulting in death.1,2 Common signs and symptoms include acute joint pain, edema, and decreased range of motion, similar to the findings of an acute rotator cuff tear or calcific tendinitis. Erythema, fever, and general malaise may also be present and are key distinguishing factors from other sources of acute pain. Additionally, there are well-documented risk factors for septic arthritis including age over 80, diabetes, prosthetic joint, joint surgery, skin infection, intravenous drug use, alcoholism, prior joint injection, rheumatoid arthritis, and other joint pathology.1,3,4 In the absence of risk factors, glenohumeral septic arthritis is rare as it is more commonly observed in an older, immunocompromised population.5-10 Here we present a curious case of a woman who experienced an acute onset of shoulder pain with confirmed septic arthritis of her native shoulder in the absence of any risk factors.
A 62-year-old female presented with eight days of diffuse, achy right shoulder pain that developed during what she described as vigorous gardening. She experienced a gradual increase in pain, with subsequent progressive loss of motion.
In the week prior to presentation to the orthopaedic surgery service, an ultrasound-guided aspiration and complete blood count (CBC) were ordered by her primary care provider (PCP). The CBC revealed a white blood cell (WBC) count of 11.39 x 109/L. When the patient did not attend the aspiration appointment and returned for follow-up with her PCP six days later with worsening symptoms, an urgent referral to orthopedics was scheduled.
On presentation to the orthopedist, she was afebrile with stable vital signs. Range of motion and strength of the shoulder were severely limited due to pain. The shoulder was edematous, but no erythema was appreciated and the extremity was neurovascularly intact. She reported no prior shoulder injuries or operations. Her medical history was notable for osteoarthritis of the knees and hips status post bilateral hip replacements. She was not on immunosuppressive medications.
Radiographs revealed no significant pathology, with slight depression of the humeral head (Figure 1). The patient underwent a magnetic resonance arthrogram (MRA) to evaluate for rotator cuff pathology, during which time simultaneous joint aspiration was performed. Initial attempt at aspiration did not reveal any fluid, so the procedure continued with contrast injection. Upon re-aspiration, nine milliliters of cloudy fluid were obtained that grew methicillin-susceptible Staphylococcus aureus (MSSA) within forty-eight hours. Consistent with septic arthritis, the MRA revealed joint debris, edema in the musculature, and abnormal signal in the subcoracoid bursa. Tears of the supraspinatus, infraspinatus, and subscapularis muscles were also identified (Figure 2).
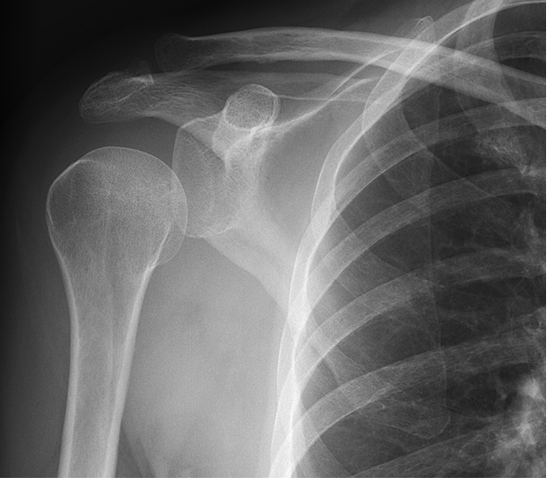
No significant pathology demonstrated. However, it was noted that the humeral head appeared depressed, indicative of an effusion.
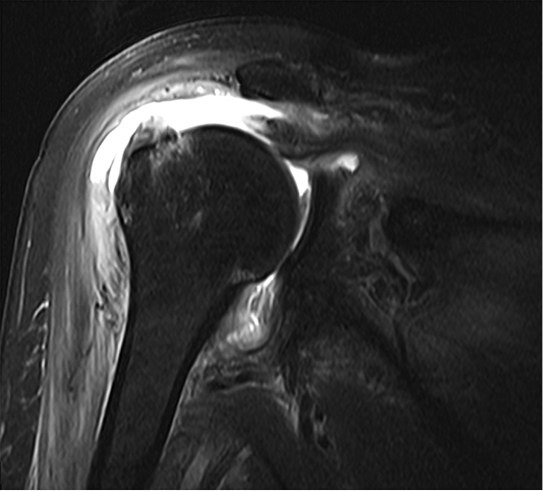
Joint debris, heterogeneous signal in the subcoracoid bursa, and edema was also noted. A coronal T2-FS sequence image is presented here.
Once cultures returned, the patient was immediately started on oral cefalexin and admitted for arthroscopic irrigation and debridement three days after the MRA. Findings at the time of surgery included diffuse synovitis and fibrinous debris within the joint, subacromial synovitis, high grade tearing of the biceps tendon, and a large rotator cuff tear. Irrigation and debridement, synovectomy, biceps tenotomy, and subacromial bursectomy were performed.
The following day, her WBC count was 11.27K/uL and blood cultures demonstrated no growth at five days. Labs two days after surgery revealed a C-reactive protein (CRP) of 111.9 mg/L, erythrocyte sedimentation rate (ESR) greater than 140 mm/h, and WBC count declined to 8.96K/uL. She was discharged with a PICC line and received six weeks of penicillin, followed by ten days of oral cefalexin. Labs trended down and were normal at two months with the exception of a mildly elevated ESR of 23 mm/h. Approximately 3.5 months after surgery the patient was doing well, although reporting lingering weakness that was attributed to her massive rotator cuff tear.
In this case presentation, the onset of pain and loss of motion were initially concerning for rotator cuff injury, adhesive capsulitis, or calcific tendinitis, though symptoms seemed out of proportion. Joint pain (85% sensitivity) and swelling (78% sensitivity) are the most sensitive indicators of septic arthritis, though they lack specificity as they give rise to a large differential of intra- and extra-articular pathologies.3,4 However, with an elevated WBC count, septic arthritis could not be excluded despite the patient’s lack of risk factors. Of note, the patient remained afebrile throughout the incident. This is not unusual, as Cleeman et al. and Duncan et al. reported fever in just 17% and 42% of patients with glenohumeral septic arthritis, respectively.6,8
Fluid culture and gram stain remain the gold standard for diagnosing septic arthritis;1,4 therefore, arthrocentesis was performed with MRA so appropriate treatment could be initiated if indicated. For septic arthritis in general, Weston et al. identified positive synovial fluid cultures in 67% of cases.2 In cases of septic glenohumeral arthritis, positive synovial fluid cultures have been reported in 68% to 89% of cases.9,8 Further synovial fluid analysis, such as cell count with differential and crystal examination, may be helpful in differentiating infectious and crystal-induced arthropathies.1,3,4 Higher WBC counts and polymorphonuclear cells over 90% are suggestive of septic arthritis.3,4 The average reported synovial WBC count in glenohumeral septic arthritis ranges from 110,9885 to 138,8006. Due to a low suspicion for infection in this case, these tests were not conducted as the results would not have changed management without supporting cultures.
ESR, CRP, and peripheral WBC count are typically elevated in septic arthritis, but they lack specificity and are most useful as a way to measure effectiveness of treatment by analyzing their trends.1 Previous reports on glenohumeral septic arthritis note average ranges of WBC count from 10.5 to 13 x 109/L, ESR from 66 mm/h to 98 mm/hr, and CRP from 83 mg/L to 134 mg/L.5,6,8,10 Imaging is seldom helpful as radiographs are often normal, while MRI may show non-specific inflammation.1,3 Depression of the humeral head, consistent with an effusion, was evident on radiographs here. Contrarily, rotator cuff tears typically result in superior translation of the humeral head.11
The etiology of infection in this patient remains unknown, but was likely hematologic. Despite a hematogenous origin, blood cultures may remain negative as Weston et al. reported positive synovial fluid and blood cultures in only 24% of cases of septic arthritis.2 Hematogenous spread is most likely in immunocompromised patients.1 Alternate routes include direct inoculation or spread of local infection,1,4 however the patient did not report trauma and there was no indication of soft tissue infection or osteomyelitis. In their series, Cleeman et al. noted that only 52% of patients with glenohumeral septic arthritis had an alternate source of infection.6 Despite a lack of reported source of infection, the infectious disease team did not deem further workup necessary in this case.
Treatment for septic arthritis entails broad antibiotic coverage after appropriate cultures are obtained, with targeted treatment once positive cultures with sensitivities are reported.1,4 Of the non-gonococcal pathogens, staphylococcus is the most common cause of septic arthritis. This is followed by streptococcus, with other bacteria characteristic of certain populations.1,2,4 This is consistent with reports specific to glenohumeral septic arthritis that identify staphylococcus as the most commonly found pathogen.5-7,9,10
Immediate irrigation and debridement is crucial, with both open and arthroscopic approaches accepted.1,5,8 Biceps tenotomy was also performed in this case as a high grade tendon tear was identified intraoperatively, and if left intact may have provided a nidus for continued bacterial growth. The reoperation rate for septic arthritis of the shoulder after initial arthroscopic debridement has been reported at 26%9 and 32%5.
Interestingly, the patient did not report shoulder weakness or pain prior to the initial onset of symptoms while gardening. This raises the question of the relationship between the patient’s rotator cuff tear and infected shoulder. The destructive and inflammatory processes associated with septic arthritis may have resulted in an acute tear, or extended a chronic tear. Alternatively, she may have first sustained a rotator cuff tear that predisposed her to infection. Other reports have noted the increased incidence of the rotator cuff tears with septic arthritis, however the exact relationship remains unclear.7-9 Not surprisingly, patients with intact rotator cuffs likely have better functional outcomes, though whether this reaches statistical significance has not been reported.8-10
To our knowledge, there are only two prior case reports of spontaneous septic arthritis in the native shoulder of a patient lacking risk factors.12,13 Griffin et al. identified a 54-year-old female with shoulder pain and decreased range of motion. Although well at presentation, within a few hours she became febrile with an elevated WBC count, CRP, and ESR. Cultures were positive for MSSA, requiring two operations for irrigation and debridement. She was discharged on intravenous antibiotics without follow-up reported.12 Hotuno et al. reported on a healthy 47-year-old male with bilateral glenohumeral septic arthritis. In addition to pain and decreased range of motion, he presented with fever, chills, tachycardia, and a systolic blood pressure of 90 mmHg. WBC count and CRP were both elevated, with aspirate and blood cultures positive for Staphylococcus aureus. Five months after two operations and a course of antibiotics, he regained almost full motion.13
Other studies have documented spontaneous septic arthritis of the native shoulder treated surgically, however they include some patients with comorbidities and risk factors. In a series by Abdel et al., of fifty native shoulder infections treated arthroscopically at an average age of 66-years-old, 57% of patients were immunocompromised, 8% had a prior injection, and 4% had prior surgery. Mortality at one year was 17%.5 Duncan et al. reported on nineteen cases of primary glenohumeral septic arthritis, with comorbidities noted in 47% of patients and an average age of 75.5-years-old.8 In a report by Jeon et al. on nineteen patients at an average age of 59-years-old with septic arthritis of the shoulder, 68% had an underlying illness and 79% had prior needle insertion. UCLA score was 26 at average 16.4 months follow-up.9 Klinger et al. discussed twenty-three infected shoulders (twenty-one patients) that were treated with surgery. Patients were an average age of 63.7-years-old, 35% had a prior injection, and 65% had an underlying disease. At average 35.3 months follow-up, pain had improved from 8.1 to 2.9 out of 10 and Constant score was 73.10 Cleeman et al. reported on twenty-two patients (twenty-three shoulders) with glenohumeral septic arthritis. In their cohort, the average age was 61-years-old, 87% had additional comorbidities, and 17% of patients had an injection. Unlike the previously mentioned studies where all patients were treated surgically, 48% in this study underwent repeat aspirations.6 Similarly, Leslie et al. reported on eighteen patients with glenohumeral septic arthritis, of which three patients were treated with aspiration while the rest received operative management. At an average age of 65-years-old, 94% of the patients had a serious comorbidity and 44% had prior needle insertion. Ten patients had a poor outcome defined as either death or no active motion at average 2.6 year follow-up.7
In conclusion, we describe the case of an otherwise healthy patient presenting with shoulder pain and weakness who was found to have septic arthritis of her native shoulder in the absence of any known risk factors. Initial key findings were pain out of proportion to alternate diagnoses, a radiographically evident effusion, and a slightly elevated WBC count. Though rare, septic arthritis should be considered in similar cases in order to avoid serious and potentially fatal consequences. While the documented risk factors do increase the likelihood of infection, their absence does not exclude it.3





